- Use: Designed for preparation and ligature.
- Material: Stainless Steel.
- Size: 140mm or 6 inches.
- Category: Plastic Surgery Instruments.
- Quality: High-quality products comply with ISO and CE standards.
- Brand: ISAHA Medical.
$325.00
ISAHA Medical’s handmade Abdominoplasty Set is equipped with 22 expertly designed instruments for the perfect tummy tuck surgery.
Features of Abdominoplasty tummy tuck Set:
- High-quality stainless steel. Free from rust and contamination.
- This comprehensive quality-assured set ensures efficiency and accuracy in abdominoplasty surgeries, making it a top choice for surgeons.
Description
Following features of ISAHA Medical’s Abdominoplasty tummy tuck Set:
- Customization of the products
Customization option is available in all of ISAHA Medical’s product range.
- Product Assurance
All of the instruments or tools manufactured by ISAHA Medical are precisely quality control checked and tested. Therefore, we assure you that all our simulated Abdominoplasty sets to suit their intended use.
- Technical Drawing
Do you want to evaluate our technical Drawing of plastic surgery sets? If yes! Please get in touch with us. So, we will send it to you for your evaluation.
Note: If you didn’t find your required items on the website, please follow the ONLINE CATALOG link or contact us via WhatsApp.
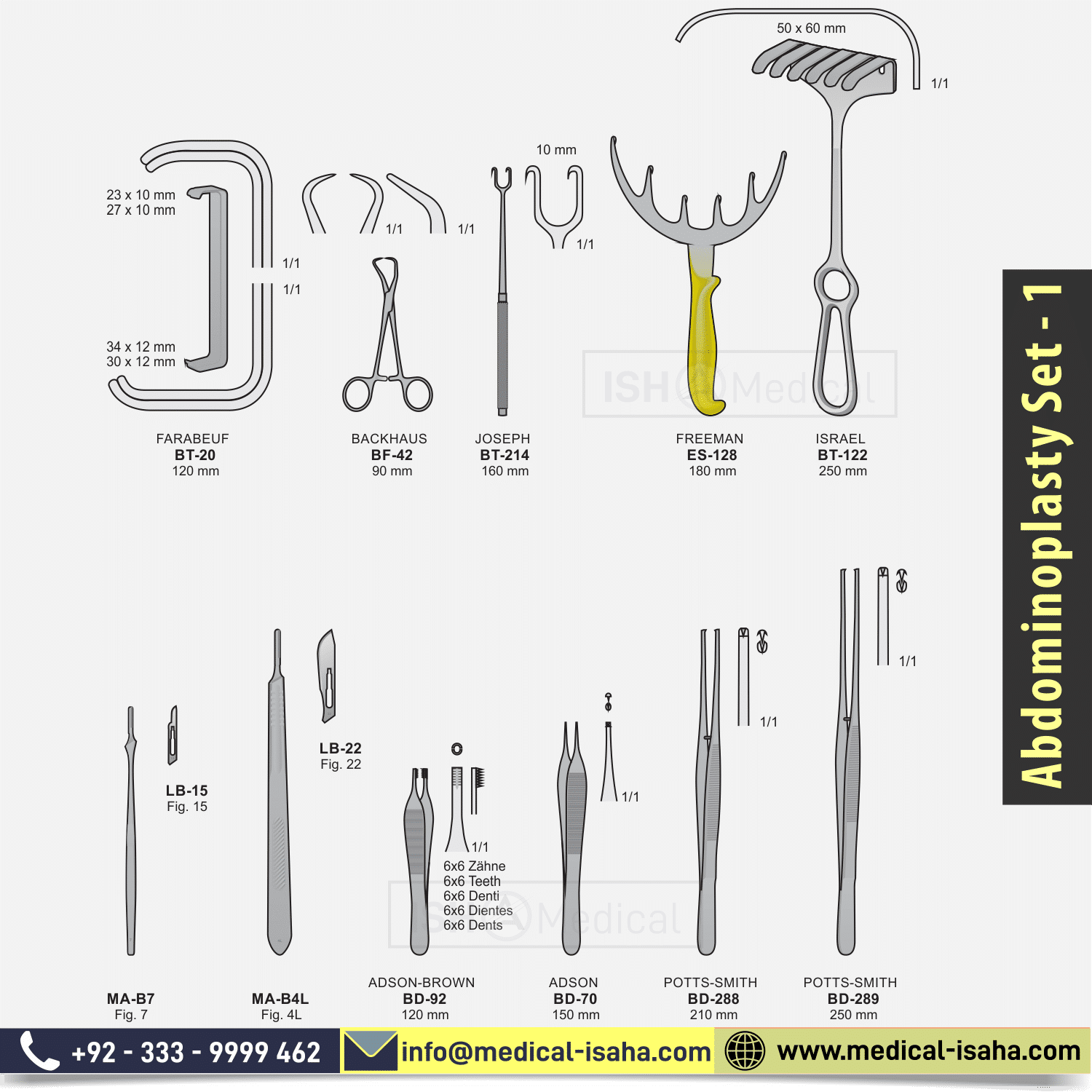
The abdominoplasty Set Includes:
-
- ADSON SURGICAL TWEEZERS cm.15
- TWEEZERS ADSON-BROWN 6×6 TEETH STRAIGHT cm.12
- POTTS-SMITH TWEEZERS STRAIGHT 1×2 TEETH cm.21
- POTTS-SMITH TWEEZERS STRAIGHT 1×2 TEETH cm.25
- BACKHAUS cm.9
- SMALL FARABEUF RETRACTOR cm.12
- ISRAEL RETRACTOR mm.50×60 cm.25
- JOSEPH HOOK 2 SHARP TEETH mm.10 cm.16
- FREEMAN RETRACTOR 6 TEETH cm.18
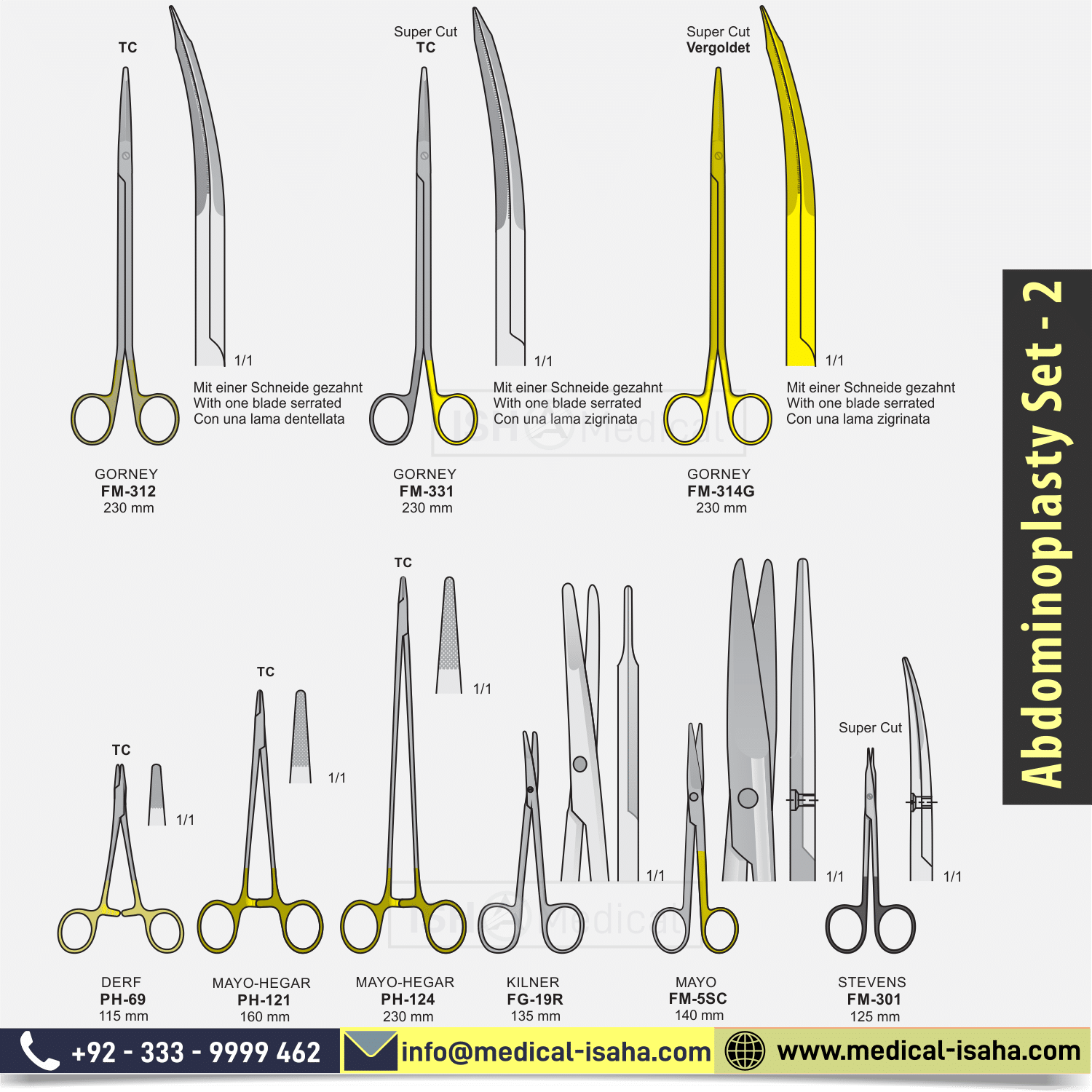
- KILNER SCISSORS STRAIGHT cm.13
- STRAIGHT MAYO SCISSORS Super-Cut cm.14
- STEVENS’S SCISSORS CURVED AND NOTCHED. Super-Cut cm.12.5
- GORNEY CURVED SERRATED SCISSORS TC cm.23
- GORNEY CURVED SERRATED SCISSORS Super Cut Gold cm.23
- CURVED GORNEY SCISSORS Super Cut TC cm.23
- BLADES FOR SCALPEL-STERILE N°15.
- BLADES FOR SCALPEL-STERILE N°22.
- STRAIGHT SCALPEL HANDLE # 4 L
- SCALPEL BLADE HOLDER HANDLE # 7 cm.16.5
- NEEDLE HOLDER DERF TC cm.12
- MAYO-HEGAR NEEDLE HOLDER, TC cm.16
- MAYO-HEGAR NEEDLE HOLDER, TC cm.23
Product Specifications of tummy tuck instruments set:
- Material: Stainless Steel.
- Sterilization Process: Not Applicable.
- BSE/TSE Statement: All Products are not made from animal origin and do not include, utilize, or involve in any production stage.
- Non-Pyrogenic standard: Not Applicable.
- DNase & RNase Free standard: Not Applicable.
- Cytotoxicity standard: Not Applicable.
- Latex-Free Statement: All our manufactured products or packaging are latex-free.
- REACH Statement: Our manufactured products do not contain the Substance of Very High Concern (SVHC). Also, they comply with the
- European Union’s Regulation (EC) No. 1907/2006 about the Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH).
- Storage Condition: Keep dry place, and avoid direct sunlight.
- Expiration Date: 5 years from the date that the product was made.
Additional information
| Brand | ISAHA Medical |
|---|---|
| Intended Use | The Abdominoplasty set contains all the necessary tools while performing the abdominoplasty procedure. |
| itemCondition | New |
| Material Type | Stainless Steel |
| Country of Origin | PAKISTAN |
| QC Testing | Following tests performs as per procedure: |
| Quality System Compliance | ISAHA Medical makes instruments for plastic surgery that meet ISO 9001:2015, ISO13485:2016, and F-899 standards. During production, plastic surgery sets are checked and controlled based on the product needs and standard operating procedures. |
| Quality Inspection | During the final quality check of Plastic Surgery Sets, we do take the following steps: – Identifying the product: Check the sample or technical Drawing visually. |
| Product's Declaration | We guarantee that the Plastic Surgery instruments we send to our valued customers as "OEM" are made of medical-grade stainless steel. Also, they meet ISO 7153-1 and F 899-20 standards. In the same way, our products are made according to the ISO 9001:2015, ISO13485:2016, and CE standards. In addition, they fall under Class 1 medical devices that meet the essential requirements of Medical Device Directive 93/42/EEC as changed by 2007/47/EC. |







Kathrin James –
Ideal set, containing all the necessary tools for cosmetic surgery of the abdomen.